White is Purity,
White is Purity,
White is Christmas,
White is Holiness,
White is Peace,
White is Coldness,
White is Brightness,
White is Cleanliness,
White is Emptiness,
White is Wholeness,
White is Newness,
White is Surrender,
White is Complete.
____________________
White, Light and Sight
White is a color perceived in the human eye by light that stimulates all three types of our color sensitive, retinal, cone cells in nearly equal amounts.
These cells individually sense the red, green and blue light that comes into our eyes, sending those signals to our brain to be interpreted.
That light may come from a light source itself (like the sun) or by reflection from object (like the light from a bright moon).
.
A closeup depiction of a retinal cone cell would look like this:
.
__________________________________
.
But, is White really a Color?
Well that may depend on whether you’re talking about
Light (Additive Color Theory) or Pigment (Subtractive Color Theory)
In Additive color theory – white is the combination of
red, green and blue – the Light Primaries.
If we were to shine white light through a prism, or water droplets, the light
would be broken down into its colored components – a full rainbow of color.
In Subtractive color theory, the combination of
magenta, cyan and yellow (the Pigment Primaries), gives the color black.
Here white is the absence of color.
Notice that the light secondary colors are the pigment’s primaries and vice versa.
__________
Some Fun with Light
You can play with variations in light color by mixing the three primary light hues – red, blue and green in the attached animated program. Once at the site, clicking on the “Run Now” button will allow you can play with different light color mixes and see how the brain observes it. See if you can make the color white with different light color combinations.
.
We see the color white in a wide variety
of Life situations. Here is just a sampling:
.
Natural White:
The non-living, inorganic chemical make-up of our teeth and bones is primarily composed of a mineral called hydroxyapatite.
>
[The Chemistry of Hydroxyapatite – Link]
.
The chemical formula for hydroxyapatite is Ca5(PO4)3(OH), but is usually written Ca10(PO4)6(OH)2 to indicate that the crystal structure has 2 units.
[Growth of Hydroxyapatite Crystals – Link]
Link to video of Hydroxyapatite synthesis below…
..
.
The structure of our teeth has an enamel coating composed of this hard hydroxyapatite mineral.
.
.
.
.
If we were to take a closer look at crystalline hydroxyapatite, it would appear as slender needles.
.
.
..
.
.
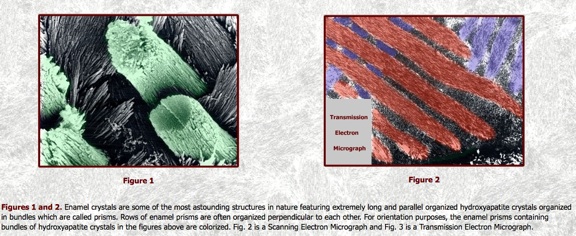
Venturing an even closer look through a transmission electron microscope, the crystalline structure of our tooth enamel would look like the following photos.
These Calcium hydroxyapatite crystals might be formed in the body
as shown in the following slide.
Now that’s something White we can
all smile about!
.
Another source of hydroxyapatite is
sea coral and it has found some very interesting uses.
Here it is used in bone enhancement ——->
Some other practical uses are listed below with video or technical links…
.
.
Injection Laryngoplasty with Hydroxyapatite (HA)
Synthesis of nanocrystalline hydroxyapatite in vitro.
HA used for Orbital Eye Implant
Hydroxyapatite Ceramic Glue for Bone Repair
Hydroxyapatite can have a negative impact on the body also.
Here are some articles on what is called Hydroxyapatite crystal (deposition) disease (HADD):
Article on “Calcific Tendonitis“
.
_________________________
.
But how do we get things white?
Well, if they’re already colored, the standard method of removing the color is by bleaching. There are multiple ways of bleaching out color. The most common is by using ordinary household bleach or sodium hypochlorite.
Bleach is an oxidizing agent that generally breaks down a portion of the molecule that causes color. There are other oxidizing agents, such as peroxide, which can also remove color, but it is not nearly as strong or harmful as hypochlorite.
Here is an example of the chemical change
bleach can cause on a very interesting molecule :
Cyanidin is a natural dye in poppies and cornflowers. The reason poppies are red while cornflowers are blue has to do with the pH of their sap. In acidic poppy sap, all OH groups in cyanidin are protonated [contain a hydrogen(H) attached to the oxygen(O)] and the light absorbed is of a higher-energy (blue); therefore, the reflected light we see is red. In basic cornflower sap, one of the OH groups is deprotonated (no H) and the molecule’s absorption shifts to green and red, and the reflected light we see is blue.

Cyanidin in poppies – (acidic) Red Cyanidin in cornflowers – (basic) Blue
Source: Foxe and Whitesell, Organic Chemistry, 2d Ed., Jones & Bartlett 1997
When dyes are oxidized (by bleach, for example), the conjugation in the molecule that causes the color, is disrupted. This disruption of conjugation raises the promotion energy of the dye, and the reflected light shifts toward the blue end of the visible spectrum.
![]() Lower Energy . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . Higher Energy
Lower Energy . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . Higher Energy
Intense bleaching whitens clothes because it shifts light absorption all the way to the ultraviolet. One possible reaction of cyanidin with bleach is shown below..
___________________________
On the other hand, if we just want white paint
or to whiten something that’s maybe a little dull,
the most common chemical used is Titanium dioxide, TiO2.
/
/
/
/
/.
.
_____________________________
And of course you can’t have a White page without including
the “White Pages”
. . . . . . . . . ..